What Is the Molecular Formula of Water? The Simple Answer Behind Earth’s Master Molecule
What Is the Molecular Formula of Water? The Simple Answer Behind Earth’s Master Molecule
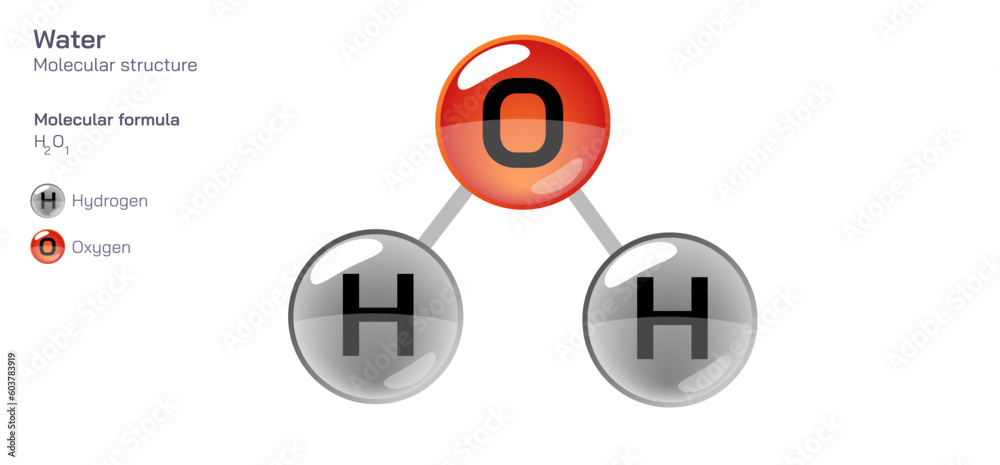
Water, the silent architect of life, governs ecosystems, shapes climates, and sustains every biological process—from the tiniest microbe to the largest mammal. At the heart of its remarkable properties lies a deceptively simple molecular formula: H₂O. This triatomic structure—two hydrogen atoms covalently bonded to one oxygen atom—underlies water’s universal role as a solvent, a temperature regulator, and a cornerstone of Earth’s hydrological cycle.
Yet, beneath this elementary formula beats the complexity of chemistry, physics, and biology fused into one essential molecule.
The Elemental Architecture: Hydrogen, Oxygen, and the Birth of H₂O
Each water molecule forms through a covalent bond, in which the oxygen atom shares electrons with hydrogen atoms to achieve stable electron configurations. Oxygen, with six valence electrons, attracts two hydrogen atoms—each contributing one—resulting in two shared electron pairs.This arrangement yields bent molecular geometry, with a tetrahedral electron domain and an observed V-shape due to repulsion between bonding pairs. The molecular formula H₂O thus reflects precise atomic ratios: two hydrogen atoms for every one oxygen, a balance refined through quantum chemistry to maximize stability and solvent efficiency. As 『Journal of Physical Chemistry』 notes, “The H₂O structure enables water’s unique polarity and hydrogen bonding, distinguishing it from other small molecules.”
Why Water’s Formula Dominates Earth’s Systems
The simplicity of H₂O belies profound functional complexity.Water’s polarity—defines by the dipole moment from oxygen’s electronegativity outweighing hydrogen—fuels its ability to dissolve salts, sugars, and proteins, making it nature’s premier solvent. This property supports nutrient transport in organisms, weathering of rocks, and glacial flow. Moreover, the hydrogen bonds linking water molecules confer high specific heat and heat of vaporization, enabling natural temperature buffers across oceans, soils, and atmospheres.
“Water’s molecular architecture creates a physical framework that stabilizes life,” explains hydrologist Dr. Elena Marquez. “Without H₂O’s simple yet precise design, Earth’s climate and biosphere would collapse.”
Bonding Dynamics: Hydrogen Links and Their Far-Reaching Consequences The three bonds in a water molecule—two O–H covalent and two lone-pair repulsion-driven hydrogen angles—give rise to unique intermolecular forces.
Hydrogen bonding, though weaker than covalent ties, is critical: each molecule interacts with up to four neighbors, forming a dynamic network in liquid water. These bonds break and reform constantly, allowing fluidity without sacrificing cohesiveness. This behavior explains why water remains liquid across a wide temperature range, essential for regulating planetary processes.
The molecular formula H₂O thus encapsulates more than atoms: it encodes a dynamic balance of strength and adaptability.
From Lab to Life: The Ubiquity of H₂O in Natural and Human Systems From microscopic reactions in industrial catalysis to macroscopic floods shaping continents, water’s H₂O blueprint persists in every drop. Microbes rely on it for metabolic reactions.
Plants depend on its polarity for vascular transport. Animals, including humans, depend on it to regulate body temperature via evaporation and guide nerve signals through ion channels. The molecular formula appears again and again: in sweat that cools skin, in blood plasma maintaining osmotic balance, in rain nourishing forests.
“Every drop is a molecular marvel,” observes environmental scientist Dr. Raj Patel, “and every molecule upstream traces back to H₂O—the molecule that makes Earth uniquely life-supporting.”
The molecular formula of water—H₂O—is far more than a chemical notation. It is the foundation of a substance capable of sustaining, transforming, and connecting life on a planetary scale.
Its simplicity when viewed at the atomic level conceals intricate physics and biology woven into every bond. In every drop of water, the elegance of chemistry meets the grandeur of Earth’s ecosystems—proving that the simplest molecules often carry the most extraordinary power.



Related Post

Monica Calhoun Son: A Visionary Communicator Shaping Modern Media Landscape

Jordana Brewster’s marriage: A Timeless Blend of Elegance, Individuality, and Modern Romance

James Nortons Future Wife Inside The: Inside the Danish Sin Derek’s Death Tapestry

Loni Wilson A Comprehensive Look At The Life And Career Of A Rising Star

