What Is Equivalence Point in Titration? The Science of Reaction Completion
What Is Equivalence Point in Titration? The Science of Reaction Completion
Equivalence point in titration marks the precise moment when the moles of titrant added exactly match the moles of analyte in solution, completing the chemical reaction with perfect stoichiometric balance. This pivotal threshold serves as the quantitative cornerstone in analytical chemistry, enabling accurate determination of unknown concentrations in samples ranging from environmental water to pharmaceuticals. Understanding this point transforms abstract titration curves into precise measurement tools, bridging theory and real-world application.
At the heart of titration lies a fundamental principle: consistency in reaction completion.
The equivalence point is not merely a statistical midpoint but a hard chemical milestone where reactants consume each other completely. For acid-base titrations, it corresponds to the pH where proton transfer is balanced; for redox reactions, it signals full electron exchange. This clarity demarcates the end of additive capacity, beyond which any further addition of titrant alters solution composition irreversibly.
The Stoichiometric Foundation of Equivalence
The significance of equivalence point rests on stoichiometric precision.
Chemical reactions follow balanced equations where reactants interconvert in fixed molar ratios—expressed by coefficients in balanced formulas. When titration progresses, reactants deplete in these ratios until exactly matched. At this junction, the calculated relationship between analyte and titrant reveals concentration, identity, and purity.
As explained by professor Elena Rodriguez, “Understanding equivalence demands grasping both mass balance and molar arithmetic—titration is chemistry in operation.”
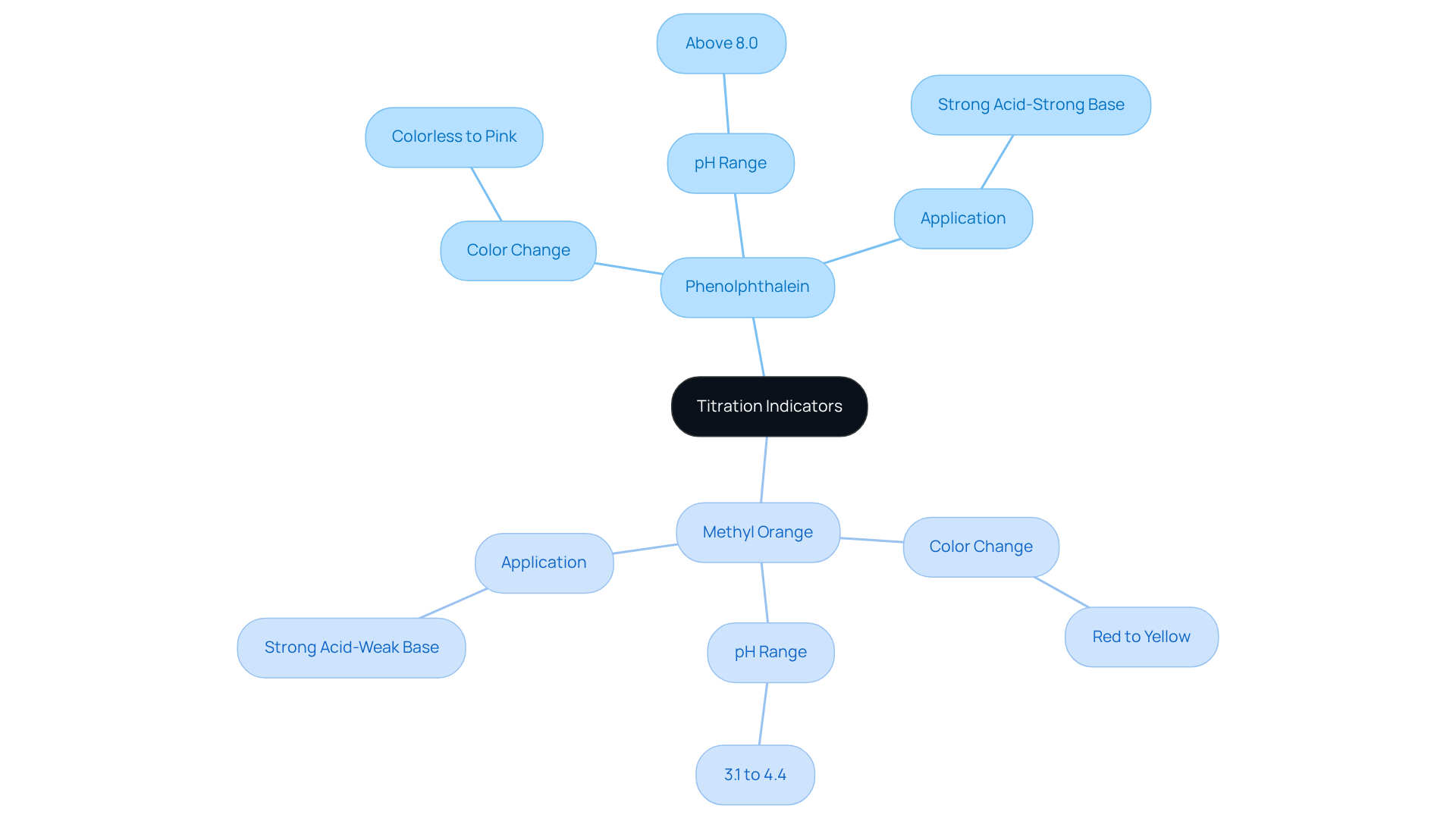
For every dip in time-based curves, every steep slope shift, equivalence anchors data to molecular scale, enabling reliable quantification. Without precise knowledge of when reactants fully react, accuracy collapses. This is why calibration with standard solutions and endpoint detection techniques like pH meters or indicators hinge on identifying equivalence point with high fidelity.
Identifying Equivalence: From Curves to Real-World Signals
Equivalence point manifests in titration curves through distinct mathematical signatures.
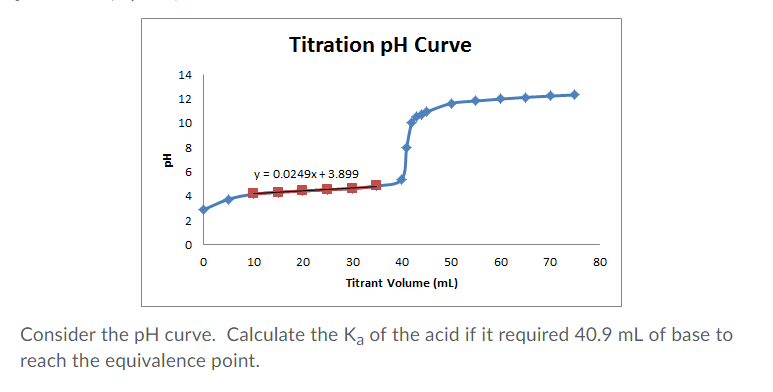
In acid-base systems, pH rises sharply at neutralization; its inflection point aligns with equivalence, though actual pH depends on acid and base strength. For strong acid-strong base reactions, equivalence occurs exactly at pH 7. In weakened acid-strong base titrations, equivalence yields basic pH (>7), while weak acid-strong base shifts the equivalence to slightly basic values—each revealing spectator ions and hydrolysis states.
Redox titrations offer additional cues: oxidation potentials shift dramatically at equivalence.
In permanganate titrations, the deep purple MnO₄⁻ fades to pale pink at endpoint, signaling complete reduction, which parallels equivalence. Similarly, iodometric titrations turn yellow-blue to clear, marking complete iodide consumption. “Each system leverages unique chemical fingerprints—pH, color, conductivity—to pinpoint completion,” notes chemist Dr.
James Wu—“finishing a reaction isn’t just about volume, but recognizing the chemistry behind the change.”
Practical Implications and Analytical Limits
Identifying equivalence with precision demands careful methodology. Errors arise from delayed endpoint detection, indicator misuse, or incomplete mixing. In industrial and lab settings, automated titrators reduce human error but require calibration against known standards.
The margin for error shrinks at low analyte concentrations, where subtle stoichiometric mismatches yield significant analytical inaccuracies. For instance, in pharmaceutical quality control, a 0.1% deviation at equivalence may invalidate drug batches. Thus, equivalence point definition transcends theory—it drives reliability in regulated environments.
Modern advancements, such as potentiometric titration and spectroscopic monitoring, refine detection by continuously tracking solution properties, eliminating reliance on colored indicators.
These tools excel in complex matrices, where visual endpoint cues fail. The convergence of instrumentation and stoichiometric rigor ensures equivalence point determination evolves alongside analytical needs.
The Broader Impact on Chemistry and Industry
Equivalence point isn’t confined to academic exercises; it underpins commerce, environmental science, and clinical diagnostics. In water treatment, precise titration quantifies pollutant removal efficiency.
In collaboration, determining calcium hardness via EDTA titration with equivalence helps maintain scale prevention in pipes. In biochemistry, enzyme assays use equivalence to assess activity, guiding drug discovery. Each application rests on the certainty that reacted equivalents truly reflect solution content—no small feat in heterogeneous systems.
Ultimately, equivalence point embodies the marriage of theory and practice: a calculated moment of complete reaction, essential for trust in chemical measurement.
It transforms titration from a procedural step into a science of precision, where every calculated volume translates into actionable insight. In laboratories worldwide, knowing exactly when a reaction ends empowers innovation, quality, and compliance.


Related Post

Sanaa Lathan’s Dating History: A Curated Journey Through Love, Growth, and Legacy

Khris Middleton’s Girlfriend: How His Relationship Narrative Reflects Quiet Strength and Modern Romance

Who’s The Lucky Lady in Ryan Trahan’s Life? Unearthing the Truth Behind His Marital Status

Shakira & Bad Bunny’s Ojitos Lindos: A Sweet, Sovereign Collaboration That Bridges Generations