The Structure of Proton Charge: Unlocking the Secrets of Atomic Electrons
The Structure of Proton Charge: Unlocking the Secrets of Atomic Electrons
At the heart of chemistry and physics lies the proton’s intricate relationship with electrons—fabricated not in isolation, but within the dynamic cloud of subatomic charge known as proton charge. This fundamental property shapes atomic stability, influences chemical reactivity, and underpins the behavior of matter across the universe. Understanding proton charge is key to unlocking the microscopic forces that govern everything from water staying liquid to stars burning through nuclear fusion.
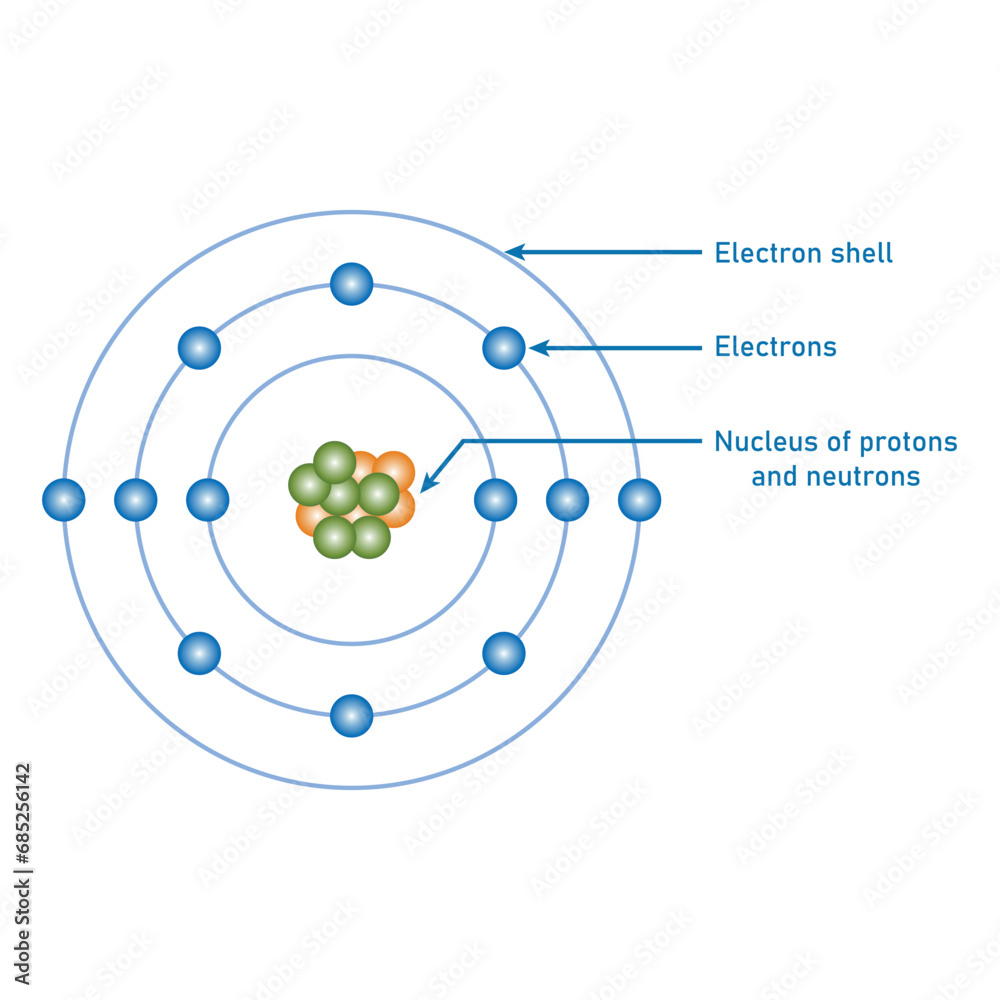
Protons, positively charged particles residing in the atomic nucleus, exert an attractive electrostatic force on orbiting electrons. This charge—the elementary charge quantified at approximately +1.602 × 10⁻¹⁹ coulombs—defines the electrostatic binding that keeps electrons in their defined energy levels. According to Coulomb’s law, the force between a proton and an electron follows an inverse-square law: as distance increases, the force weakens, but at the nanoscale, it remains potent enough to sustain atomic structure.
Electron binding energy, derived from the interplay of proton charge and electron motion, determines an atom’s chemical character. The stronger the electrostatic attraction, the more energy is required to remove an electron—giving rise to ionization energy, a fundamental metric in predictive chemistry. For example, hydrogen’s single proton attracts its lone electron with sufficient force to support life-sustaining molecular bonds, while elements with protons and electrons balanced differently exhibit vastly varied reactivity.
Measuring Proton Charge: Precision at the Quantum Level Accurately quantifying proton charge demands advanced instrumentation.
Modern techniques leverage both theoretical models and empirical data from particle accelerators. The proton’s charge was first precisely measured in the mid-20th century using ionization chambers and mass spectrometers, confirming its integer-like magnitude within experimental uncertainty. Today, atomic force microscopy (AFM) and quantum electrodynamics (QED) simulations refine these values, ensuring precision critical to quantum chemistry and materials science.
As physicist Richard Feynman once noted, “The charge of the proton is not just a number—it’s a fingerprint of electromagnetism.”
How Proton Charge Drives Atomic and Molecular Behavior
The electrostatic influence of the proton extends beyond simple attraction—it governs orbital configurations, electron spin states, and even molecular geometry. In covalent bonding, shared or unequal electron distribution responds directly to proton charge, determining molecular polarity and reactivity. In polar molecules, protons induce charge asymmetry, enabling hydrogen bonding—a key force in biological systems like DNA and proteins.Another critical aspect is spectroscopy, where proton charge affects energy transitions detectable via absorption or emission spectra.
Shifts in spectral lines, influenced by proton-electron interactions, provide remote sensing tools in astronomy and environmental monitoring. For instance, the detection of molecular clouds in space relies on identifying proton-driven dipole transitions, illuminating cosmic chemistry millions of light-years distant.
From Atomic Nuclei to Cosmic Scale: Proton Charge’s Far-Reaching Impact
Beyond individual atoms, proton charge shapes the behavior of matter at every scale—from synthetic nanomaterials to stellar cores.In fusion reactors, controlling proton proximity via electromagnetic fields seeks to replicate the nuclear fusion powering the sun, where proton-proton collisions overcome Coulomb repulsion at extreme temperatures to generate energy. Understanding these interactions hinges on accurate modeling of proton charge effects.
In materials science, proton charge influences conductivity and surface properties.
Semiconductors, essential for electronics, rely on electron-hole dynamics conditioned by atomic-level charge distributions, including proton-induced polarization effects at interfaces. Similarly, catalyst surfaces depend on proton affinities to facilitate redox reactions—key steps in industrial catalysis and clean energy storage, such as in fuel cells.
Real-World Applications and Future Frontiers
Proton charge is no longer a theoretical curiosity—it drives innovation across disciplines. In medicine, proton beam therapy—guided by precise charge calculations—delivers targeted radiation to tumors with minimal damage to surrounding tissue.Advances in quantum computing leverage proton-based qubit systems, where charge coherence is maintained through careful manipulation of electromagnetic environments. Emerging research explores proton charge effects in exotic states of matter, such as quark-gluon plasmas in high-energy physics, or quantum dots engineered for optoelectronic precision. As experimental methods grow more sensitive, the ability to “see” proton charge in action deepens, offering insights into fundamental forces and enabling breakthrough technologies rooted in atomic-scale control.
From the fragile balance of electron orbitals to the energetic engines of cosmic stars, proton charge stands as a cornerstone of modern science. Its influence permeates chemistry, material innovation, and energy technologies—proof that invisible forces shape the visible world. With continuous refinement in measurement and application, the study of proton charge remains a vital frontier, unlocking deeper truths about the building blocks of reality.


Related Post

What Does Pmo Mean

Edi Patterson & Daniel J O Connor: A Union Rooted in Irish Heritage and Family-Driven Success

Mastering Deephot Links: Unlock Deep Linking Mastery with T Dniel Pinto’s Essential Guide

Lahna Turner’s New Husband: A Transformative Chapter Defined by Love, Legacy, and Quiet Strength

