The Phosphate Backbone: Unlocking DNA’s Structural Blueprint
The Phosphate Backbone: Unlocking DNA’s Structural Blueprint
Beneath the spiraling ladder of DNA lies a critical, often overlooked hero: the phosphate backbone. This unassuming molecular chain forms the structural spine of the double helix, orchestrating the precise spatial organization that enables DNA’s vital role in genetic inheritance and cellular function. Far more than a passive scaffold, the phosphate backbone governs DNA’s conformation, stability, and interaction with proteins—laying the foundation for molecular biology’s most fundamental processes.
Unlocking its secrets reveals how life’s code maintains integrity across generations and responds dynamically to the cell’s needs.
Architecture of Order: The Phosphate Backbone as Structural Scaffold
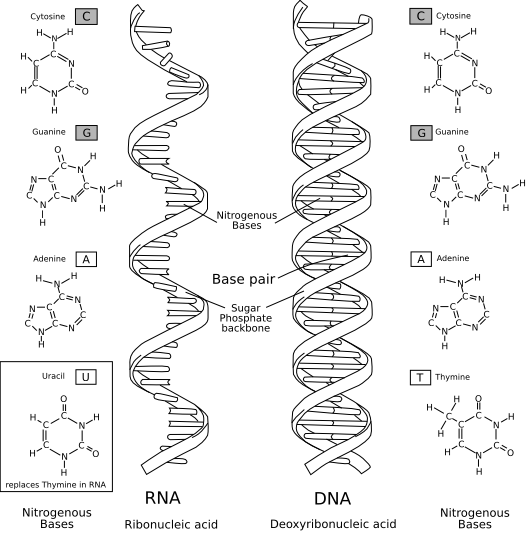
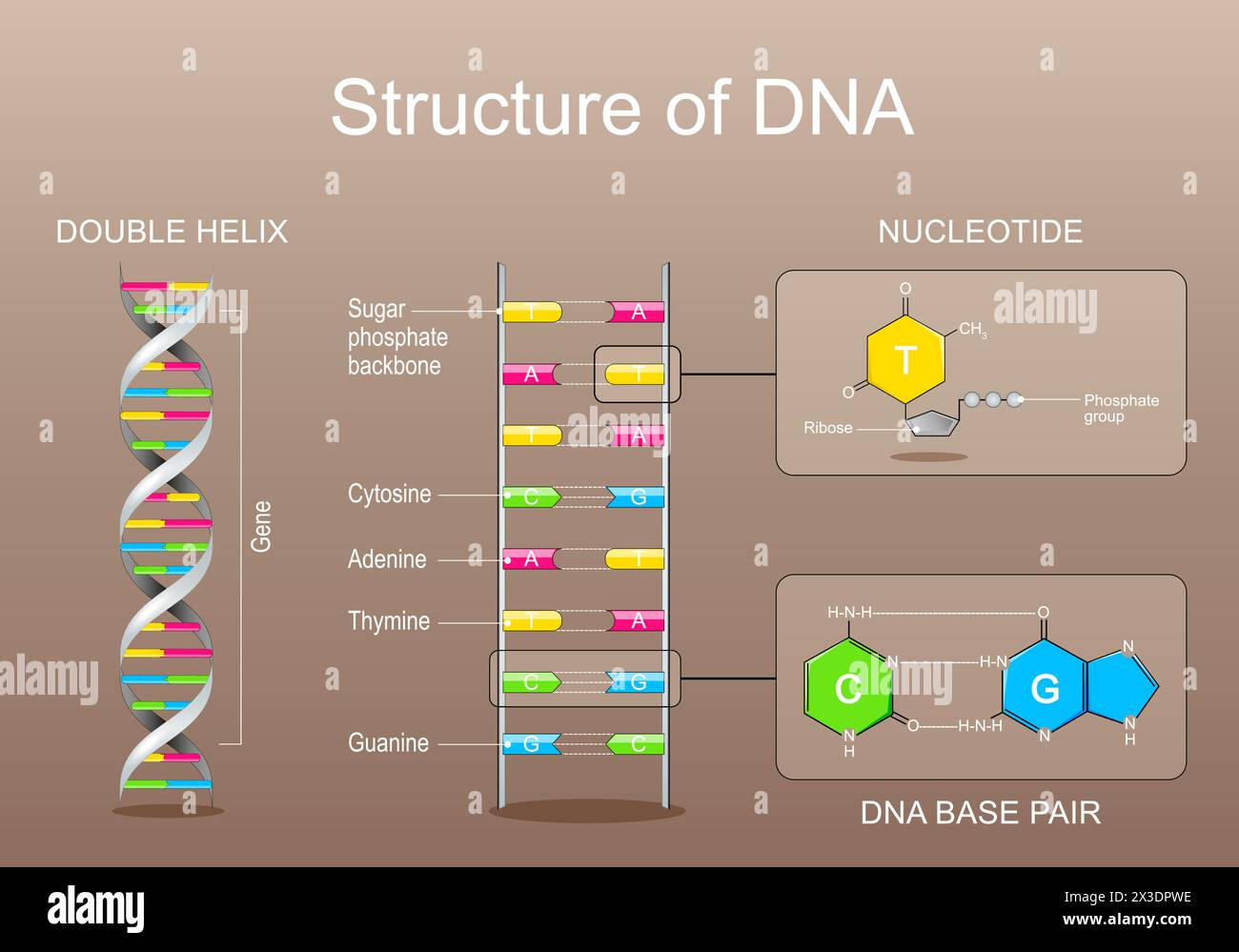
At the core of DNA’s triple-helix architecture is a repeating sugar-phosphate unit, linked end-to-end via phosphodiester bonds. Each phosphate group, derived from phosphoric acid, carries a negative charge due to its ionized hydroxyl groups.This ci>
The negatively charged phosphate groups position themselves at the exterior of the helix, creating a polarized bilateral frame that supports the sugar-phosphate backbones’ alignment. Each phosphate links two sugar molecules—deoxyribose in DNA—forming the lateral radio of the double helix. The alternating pattern of phosphate and sugar creates a rigid yet flexible framework, allowing DNA to twist into its iconic double-spiral while resisting physical strain during replication and transcription.
As biochemist Dr. Elena Torres notes, “The phosphate backbone isn’t just a connector—it’s the architect of DNA’s spatial harmony, defining the shape that underpins every biological function.”
The backbone’s geometry follows a consistent pattern: a 3.3 Å repeat distance between base pairs, translating to a stable, uniform helical rise of approximately 3.4 Å per turn. This precise molecular architecture is governed by hydrogen bonding between adjacent bases and van der Waals interactions stabilized by the phosphate’s phosphate-sugar backbone.
Without this structured rigidity, DNA would collapse into structural disarray, jeopardizing its role as the cell’s blueprint.
Charge Dynamics: Engineering DNA’s Stability and Interaction
The phosphate backbone’s predominant feature is its high negative charge density, a dual-edged sword critical to DNA’s behavior. This charge repulsion between adjacent phosphate groups generates both mechanical stress and dynamic flexibility.“Electrostatic forces within the phosphate backbone influence DNA’s ability to unwind and rewind,” explains molecular biophysicist Dr. Rajiv Mehta. “The repulsion must be counterbalanced by favorable base stacking and ionic shielding—primarily from cellular potassium and sodium ions—to maintain double-helical stability in physiological conditions.”
Sodium ions, in particular, play a vital role in neutralizing the phosphate’s charge.
In a typical cellular environment, thousands of singly charged phosphates create a strong net negative potential across the helix. Yet, the surrounding ionic milieu reduces electrostatic strain, enabling proteins like helicase and topoisomerase to efficiently manipulate the DNA structure without causing unwinding or damage. This delicate balance preserves genomic integrity during replication, repair, and gene expression.
Dynamic Conformations: Adaptability of the Backbone in Cellular Processes
DNA’s phosphate backbone is not a static structure but a dynamic player in biological transformations. During replication, helicase enzymes unwind the double helix by cleaving hydrogen bonds between base pairs, yet the backbone remains intact along the single-stranded regions. Transcription factors bind specific DNA sequences by inducing localized distortions—kinks, zippers, or bends—mediated by subtle backbone flexibility.These conformational changes allow the DNA to “flex” and expose binding sites without compromising structural continuity.
In gene regulation, methylation of cytosine bases near the phosphate backbone influences chromatin compaction by altering interaction strength with histone proteins. Methylated DNA often stiffens the backbone, promoting tighter wrapping and degraded gene expression—a mechanism central to epigenetics.
Similarly, mechanical forces from cellular processes like cytokinesis or chromosome segregation induce temporary backbone stretch and twist, signals cells use to detect structural stress and respond appropriately.
Engineered Insights: The Backbone in Biotechnology and Medicine
Understanding the phosphate backbone’s properties has revolutionized biotechnology and therapeutic development. In CRISPR-Cas9 gene editing, precise targeting depends on the DNA backbone’s geometry — Cas9’s guide RNA must correctly align with the phosphate-sugar framework to induce double-strand breaks.Similarly, DNA nanotechnology exploits the backbone’s predictable hydrogen bonding and flexibility to design intricate nanostructures for drug delivery or biosensing.
Drug design also leverages phosphate backbone knowledge. Nucleotide analogs—used in antiviral and anticancer therapies—often incorporate modified phosphates to enhance stability and binding affinity while evading cellular degradation.
Conversely, targeting abnormal phosphate interactions in disease states, such as altered chromatin packaging in cancer, presents new avenues for epigenetic drugs aiming to normalize gene expression patterns.
Quantifying Flexibility: Experimental Validation of Backbone Behavior
Modern biophysical techniques have illuminated the phosphate backbone’s nuanced behavior. X-ray crystallography and cryo-electron microscopy reveal intricate hydrogen-bond networks that stabilize sugar-phosphate repeats.Nuclear magnetic resonance (NMR) spectroscopy shows backbone dynamics on atomic scales, capturing fluctuations that enable DNA’s functional adaptability. Single-molecule force spectroscopy applies controlled tensile stress to stretches of synthetic DNA, measuring torque and displacement to quantify how the phosphate backbone resists or yields under tension.
Data from these experiments confirm that while the backbone is rigid enough to maintain structure, it exhibits localized flexibility—especially near biologically critical regions.
This dual property enables DNA to store dense genetic information while remaining accessible and responsive. “The backbone’s mechanical resilience and dynamic nature are key to DNA’s evolutionary success,” notes structural biologist Dr. Lina Chen.
“It’s the invisible wire that holds structure but allows motion.”
Implications for Synthetic Biology and Future Research
The phosphate backbone’s structural and functional centrality fuels innovations in synthetic biology. Researchers construct artificial genetic systems using synthetic backbones—modified phosphates or even non-phosphate polymers—seeking stability and programmability beyond natural constraints. Yet, preserving biological mimicry remains crucial: any synthetic structure must replicate backbone flexibility, charge distribution, and ion interaction to function predictively in cellular environments.Emerging tools like base-editing and synthetic chromatin engineering demand precise manipulation of the DNA backbone in context. Advances in computational modeling now simulate how phosphate scaffold modifications affect DNA folding, mechanical strength, and protein binding—accelerating design cycles for novel biomaterials, programmable DNA circuits, and regenerative medicine scaffolds.
In natural systems, ongoing studies probe how environmental factors—pH, ion concentration, mechanical stress—affect backbone integrity during DNA maintenance.
Understanding these dynamics promises deeper insights into DNA diseases, aging, and DNA repair mechanisms.
Unlocking Life’s Code: The Phosphate Backbone as a Key Molecular Architect
The phosphate backbone is far more than a chemical afterthought—it is the structural and dynamic foundation upon which DNA’s revolutionary blueprint rests. Its charged, repeating architecture enables the double helix’s stability, shapes interactions with proteins, and orchestrates genome-spanning processes essential to cellular life.From stabilizing replication forks to enabling epigenetic regulation, this unheralded chain guides the flow of genetic information with astonishing precision. As scientific mastery over DNA deepens, the phosphate backbone remains a central locus of discovery—unlocking not only the structural secrets of life’s molecule but also the potential for transformative advances in medicine, biotechnology, and beyond.


Related Post

Imogen Lucie: The Rising Star Redefining Contemporary Dance and Performance Art

How Carlton Fresh Prince Redefined Comedy Through Viral Lyrics and Timeless Social Commentary

Where Is Joe Buck From? Tracing the Roots of a CNN Anchor

Wolverine’s Origins Reimagined: The Chilling Power Behind the Wolverine The Origins Cast

