The Molecular Puzzle of Nitrogen Dioxide: Decoding Its Lewis Structure and Chemical Significance
The Molecular Puzzle of Nitrogen Dioxide: Decoding Its Lewis Structure and Chemical Significance
Nitrogen dioxide (NO₂) stands at the crossroads of atmospheric chemistry and molecular complexity—a seemingly simple molecule with a paradoxical nature. At its core, it embodies both stability and reactivity, structured by a Lewis framework that belies its dynamic role in environmental processes and industrial applications. Understanding NO₂’s electron distribution through Lewis structure analysis reveals far more than atomic bonding—it exposes the molecular logic behind its participation in critical chemical transformations, from ozone depletion to signal transduction in biological systems.
As scientists continue to decode its electronic architecture, NO₂ emerges not just as a pollutant, but as a pivotal player in Earth’s chemical equilibrium.
Decoding the Lewis Structure: The Electronic Architecture of NO₂
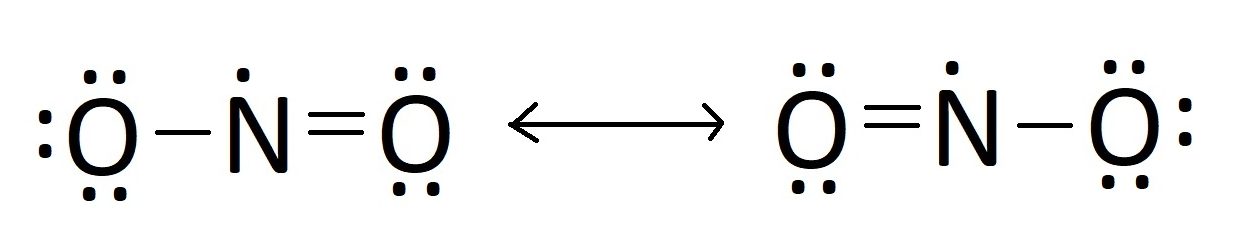
Lead by nitrogen’s central positioning, nitrogen dioxide’s Lewis structure reflects a resonance hybrid that defies static representation. Nitrogen, with five valence electrons, forms two primary bonds with oxygen atoms while retaining one unpaired electron, giving rise to a free electron pair that influences reactivity.Each oxygen atom bears seven valence electrons, sharing one bond with nitrogen and carrying a formal charge تأخ in the commonly depicted Koeppen structure. However, the true picture emerges through resonance: molecular models show NO₂ oscillating between two resonance forms, each featuring a double bond in one oxygen-lewis arrangement and a lone pair on nitrogen. This dynamic structure is captured in experimental data from spectroscopic studies, which confirm an average bond length of approximately 1.23 Å and a bent molecular geometry with a bond angle near 135°.
The electron count reveals complexity: nitrogen contributes one lone pair, oxygen atoms each contribute three non-bonding electrons. This distribution results in a molecule with 18 valence electrons—meeting the octet rule across atoms but generating a localized domain of high electron density. The unpaired electron on nitrogen imparts paramagnetic character, distinguishing NO₂ from diamagnetic nitrogen dioxide isomers.
Quantum chemical calculations, including density functional theory (DFT) models, further elucidate the energy distribution across resonance forms, showing partial bond orders of 1.5 between nitrogen and each oxygen. These mathematical representations translate directly into observed spectroscopic shifts and reactivity patterns.
Resonance défies simple depictions, yet the resulting hybrid structure stabilizes NO₂ in volatile atmospheric conditions while preserving its capacity for redox activity. The molecule’s polarity, driven by the electronegativity difference between nitrogen and oxygen, enhances its solubility in water—a key factor in its environmental impact.
Experimental evidence shows NO₂ absorbs strongly in the visible spectrum, particularly at 400–450 nm, contributing to its brownish air discoloration and serving as a diagnostic signature in monitoring air quality. Thus, the Lewis structure is not a static diagram but a living model reflecting NO₂’s adaptive chemical behavior.
Chemical Dynamics: NO₂ in Environmental and Industrial Reactions
Nitrogen dioxide’s role extends far beyond molecular curiosity—it is a reactive linchpin in atmospheric chemistry and industrial synthesis. As a potent oxidizing agent, NO₂ participates in radical chain reactions that drive tropospheric ozone formation and degradation of volatile organic compounds (VOCs).Its ability to interact with hydroxyl radicals (•OH) and emit nitrogen trioxide (NO₃) under sunlight initiates self-sustaining cycles that influence air quality across urban landscapes.
In industrial chemistry, NO₂ functions as a critical intermediate. In the production of nitric acid via the Ostwald process, NO₂ is sequentially oxidized: first converting to NO, then to NO₂, and ultimately reacting with water to yield HNO₃.
This pathway underpins the global supply of nitric acid—essential in fertilizer, explosive, and pharmaceutical manufacturing. Moreover, NO₂’s redox flexibility enables its use in catalytic converters, where it aids in the reduction of carbon monoxide (CO) and unburned hydrocarbons through complex surface reactions on platinum or rhodium catalysts. These applications illustrate how the molecule’s electronic structure enables electron transfer and bond rearrangement, making it indispensable in both natural and engineered systems.
Biologically, NO₂’s redox activity intersects with cellular signaling.
While highly toxic at high concentrations, low levels modulate cytochrome c oxidase in mitochondria, influencing cellular respiration. This duality—reactive yet regulated—highlights the precision of nature’s design. Structural studies using X-ray crystallography and electron paramagnetic resonance confirm NO₂’s binding to heme centers, reinforcing its role as a molecular messenger in oxidative stress responses.
From polluted skies to bioenergetic networks, the Lewis framework of NO₂ directly informs its multifaceted reactivity.
Safety, Regulation, and the Future of NO₂ Research
Amid its chemical utility and environmental concern, nitrogen dioxide stands at the heart of global air quality policy. Classified as a criteria air pollutant by the U.S. EPA and the World Health Organization, NO₂ exposure is linked to respiratory ailments, cardiovascular stress, and premature mortality.Regulatory agencies enforce strict limits—current U.S. standards cap ambient NO₂ at 53 parts per billion over eight hours—to mitigate health impacts. Yet, ongoing research seeks to refine these thresholds using improved atmospheric models integrating detailed bonding energetics and reaction kinetics derived from molecular simulations.
Emerging studies probe NO₂’s role in nanoscale catalysis and environmental remediation. Engineered nanostructures functionalized with nitrogen-containing groups aim to mimic NO₂’s redox behavior for targeted pollutant degradation. Computational chemists continue to refine computational models, leveraging quantum mechanical insights into electron delocalization to predict reactivity in unprecedented detail.
These advancements promise cleaner industrial processes and smarter environmental monitoring systems.
In summary, nitrogen dioxide is more than an atmospheric irritant—it is a molecular puzzle whose Lewis structure unlocks profound chemical significance. From its resonant bonding and polar character to its pivotal role in energy conversion and pollution dynamics, NO₂ exemplifies how fundamental molecular architecture drives macroscopic consequence. As research deepens, so too does our appreciation for the elegant mechanics underlying one of Earth’s most consequential molecules.
Understanding its puzzle is not just a scientific triumph, but a cornerstone for environmental stewardship and technological innovation.


Related Post

Trevor Brolin Films: Where Brutal Realism Meets Cinematic Precision
Steve Harvey Mama: The Quiet Force Shaping Modern Parenting Wisdom

Eduardo Santamarina: The Architect of Media Innovation in the Digital Age
Quotex Trading in Bangladesh: Your Ultimate Guide to Thriving in the Dynamic Local Market

