Protons, Neutrons, and Electrons: The Atomic Heartbeat of Hydrogen
Protons, Neutrons, and Electrons: The Atomic Heartbeat of Hydrogen
Amid the silent dance of subatomic particles, hydrogen—element number one of the periodic table—stands as the universe’s most fundamental storyteller. Driven by just three particles—protons, neutrons, and electrons—this simple atom reveals profound complexity beneath its unassuming surface. From the fusion powering stars to the delicate balance defining chemical identity, hydrogen’s atomic heartbeat pulses through everything.
Understanding its structure is key to unlocking not only the essence of matter but also the origins of life itself.
The Foundation: Protons, Neutrons, and the Quantum Architecture
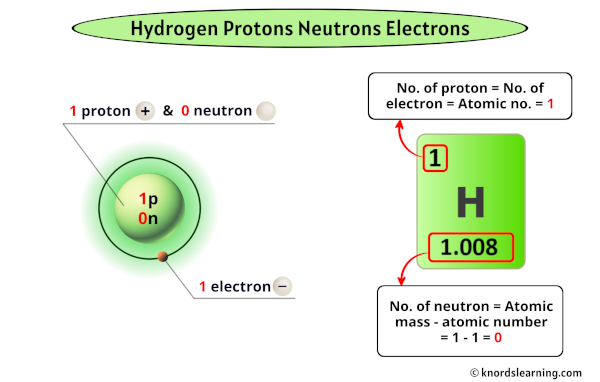
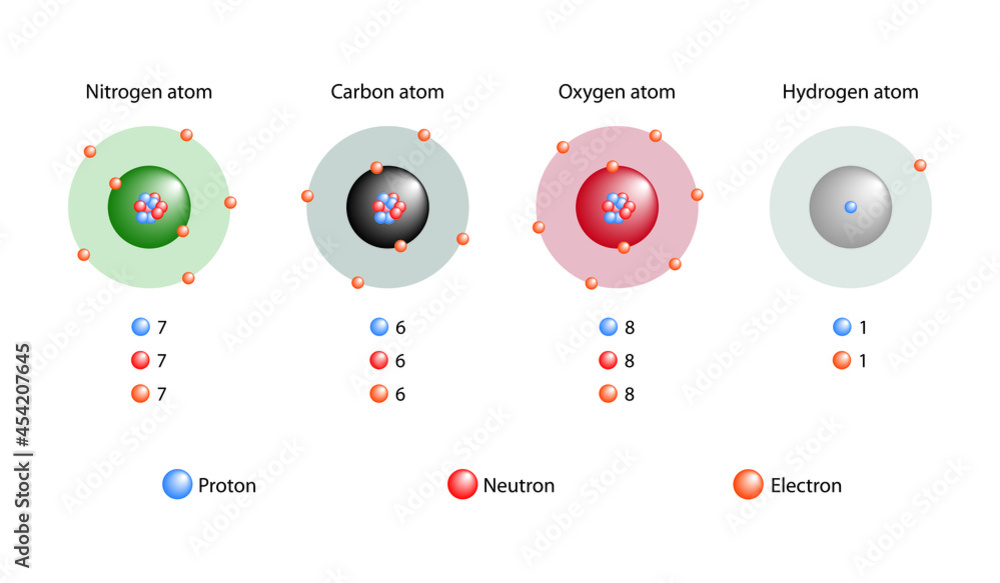
Hydrogen’s core consists of a single proton, the positively charged particle that defines its atomic number and governs its elemental identity. Unlike heavier elements with layered structures, hydrogen possesses only one proton at its nucleus—a minimalist hub upon which the atom pivots.Neutrons, electrically neutral and welded to the proton, provide critical stability. While most hydrogen atoms contain no neutrons (protium), variants like deuterium and tritium carry one and two neutrons respectively, forming isotopes with slightly altered nuclear properties. “Hydrogen’s simplicity masks its importance—its proton alone sets the stage for the diversity of chemistry in the universe,” explains Dr.
Lena Torres, nuclear physicist at the Max Planck Institute. “Yet, even here, variation matters.” In 99.98% of natural hydrogen, the nucleus holds exactly one proton and zero neutrons, a configuration so stable that it enables hydrogen to exist as the dominant fuel for stellar fusion. Without this proton anchor, the delicate balance holding matter together would collapse at the quantum level, revealing the element’s foundational role in cosmic evolution.
Neutrons, when present, lighten the atomic mass but thicken nuclear interactions. Deuterium—hydrogen with a neutron—exhibits smoother nuclear binding, a trait exploited in nuclear reactors and advanced fusion research. Tritium, with two neutrons, holds potential as a fusion fuel but also introduces complexity in handling due to its radioactivity.
Together, protons and neutrons form the nucleus, a dense, invisible core where quantum forces determine hydrogen’s physical behavior. This microscopic arrangement sets the stage for valence electrons, the dynamic players whose movements define chemical interactions.
Valence Electrons: The Silent Architects of Hydrogen’s Chemistry
Hydrogen’s single electron, loosely bound to its core, governs nearly every interaction in which the atom takes part. This lone valence electron binds with remarkable flexibility, capable of forming strong covalent bonds with other elements—a hallmark of hydrogen’s pervasive chemistry.Whether bonding with oxygen in water or attaching to carbon in organic compounds, the electron’s behavior is central. “Hydrogen’s electron is surprisingly versatile,” notes Dr. Rajiv Mehta, a quantum chemist specializing in light elements.
“It forms bonds not only with the most electronegative elements like fluorine but also in weakly interacting systems, enabling roles from hydration to energy transfer.” In molecular form, hydrogen typically exists diatomic (H₂), where two hydrogen atoms share their single electron pair in a stable covalent arrangement. However, in ionic compounds like sodium hydride or in stereochemical contexts such as ammonia, the electron behaves selectively—participating in bonding or remaining transiently available for reaction. The electron’s position also affects hydrogen’s physical state.
In solid or liquid form, its mobility contributes to unique properties like low boiling point (−252.8°C), a direct consequence of weak intermolecular forces governed by electron distribution. When ionized—gaining or losing that electron—hydrogen transforms: the resulting proton becomes a potent hydrogen ion (H⁺), fundamental in acid-base chemistry and biological processes.
This dual role—as both electron donor and acceptor—makes hydrogen indispensable in both atomic and ecological systems, from enzymatic catalysis to planetary atmospheres.
Hydrogen’s Isotopes: Subtle Variations, Profound Implications
Though nearly all hydrogen atoms feature one proton and no neutrons, natural abundance reveals isotopic travelers: protium (¹H), deuterium (²H, or D), and tritium (³H, or T).These variants differ only in neutron count, yet induce measurable shifts in behavior and stability. Protium, the common form, dominates due to its simplicity and abundance—vital in fossil fuel chemistry and biological metabolism. Deuterium, with one extra neutron, is heavier and less reactive, slowing diffusion and influencing phase changes, a phenomenon exploited in GPS satellite calibration and nuclear magnetic resonance imaging.
Tritium, radioactive and rare, decays over time but remains crucial in fusion experiments and biological tracing. “Isotopic differences aren’t just academic—they redefine how hydrogen functions in nature,” says Dr. Torres.
“Deuterium, for example, behaves like a heavier twin: it slows down hydrogen chemical reactions, which alters the kinetics of biochemical pathways and even affects climate through water isotope fractionation.” These subtle distinctions underscore how subatomic variation governs macroscopic outcomes. In oceanography, the ratio of deuterium to protium reveals ancient climate patterns. In fusion science, deuterium-tritium reactions provide the most efficient energy release known to humanity.
In medicine, tritiated compounds enable precise molecular tracking.
Thus, even an element as elementary as hydrogen demonstrates that complexity often lies beneath simplicity—defined by particles that shape everything from stars to cellular function.
In every bond formed, every reaction catalyzed, and every energy released, protons, neutrons, and electrons coalesce into a unified atomic heartbeat. Hydrogen, though simple, stands as a microcosm of the universe’s underlying order—protons affirming identity, neutrons modulating stability, and electrons igniting chemistry.
Understanding this trio is not just a journey into atomic structure; it is a recognition of the fundamental dance that makes matter, life, and energy possible.


Related Post

Devanne Villarreal: Boss, Partner, and Central Figure in a Husband-Focused Narrative

Carrie Hamilton: A Voice That Challenged Hollywood and Resonated Beyond the Screen

Is Shiri Spear Still Married? Unveiling the Truth Behind the Celebrity’s Life Status

Unlocking Sawyer Fredericks’ Bio: Age, Net Worth, Height, and the Facts Behind a Rising Star

