Does a Liquid Have a Definite Volume? The Science Behind Flowing Clarity
Does a liquid possess a definite volume, or does its flowing nature inherently defy fixed measure? At first glance, liquids may appear to conform to classical definitions—consistent volume, smooth flow, predictable behavior—but the scientific reality reveals a more nuanced picture. The interplay between molecular structure, temperature, pressure, and intermolecular forces shapes how liquids behave, raising fundamental questions about their volumetric stability.
While most everyday liquids behave as if they have a definite volume, modern physics and materials science reveal that this assumption holds only under specific conditions—beyond these, liquid behavior becomes complex and contextual. Understanding this requires examining both empirical observations and theoretical frameworks that govern liquid dynamics.
The Myth of Absolute Volume: When Liquids Defy Expectations
Contrary to intuition, liquids do not always possess a perfectly fixed volume—though most do under ordinary conditions. High-precision measurements in laboratory settings confirm that pure liquids like water or ethanol exhibit minimal, predictable volume fluctuations within a narrow range, governed by thermal expansion and compression.Yet this "definiteness" is relative, not absolute. Laboratory instruments, such as laser interferometry and high-resolution gravimetric sensors, detect volume changes down to parts per million when temperatures or pressures shift by just fractions of a degree. Thermodynamic Sensitivity - Temperature’s role: As temperature increases, molecular motion intensifies, increasing average separation between particles. This expansion reduces density and, in conventional understanding, appears to dilute volume.
- Pressure influence: Elevated pressure compresses molecules, counteracting thermal expansion and yielding a denser, more tightly packed state. In extreme environments—like deep oceans or planetary interiors—pressure can significantly alter a liquid’s effective volume. - Intermolecular forces: Hydrogen bonding in water or van der Waals forces in hydrocarbons create dynamic networks that resist compression and shape how liquid volume responds to external stress.
“Liquids are far from static,” explains Dr. Elena Rostova, a physical chemist at the Institute of Advanced Fluids, “their volume fluctuates ever so slightly with temperature, pressure, and even nearby electric fields—evidence that ‘definite volume’ is better described as operationally stable under standard conditions.”
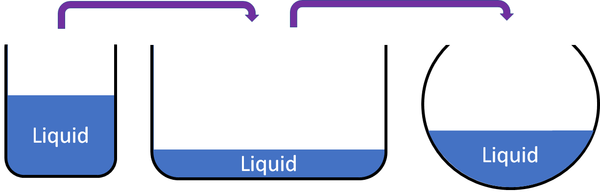
Why Liquids Appear “Definite” at Everyday Scales
Human perception and measurement limitations mask subtle variability. In day-to-day use—fill a glass, pour milk, monitor industrial tanks—volume behaves as consistent. This practical stability arises because measurement tools average fluctuations over time, and molecular motion, while significant, averages out at macroscopic scales.The human eye and conventional instruments detect only bulk changes, not atomic shifts. For most applications—instructional, industrial, or domestic—liquid volume remains functionally defined. A liter of water poured into a bottle occupies the same volume numerically in each transfer, reinforcing the illusion of definition.
Macroscopic Consistency depends on system boundaries and environmental control. Temperature fluctuations in a room, changes in atmospheric pressure, or mixing with other fluids introduce real, though often negligible, deviations. Yet in stable environments—laboratory, manufacturing, or household settings—liquids maintain effectively constant volume, making a stable definition pragmatically valid.
Beyond Macroscopy: Liquid Behavior in Extreme Conditions
When pushed beyond terrestrial norms, liquids display volumetric behavior that challenges conventional understanding. Under ultrahigh pressures—such as those found in gas giant cores or deep Earth layers—water transforms into ice phases like ice VII, compressing molecules into crystalline structures that defy conventional density expectations. This phase transition drastically reduces volume despite similar mass, illustrating how volume remains context-dependent.Similarly, supercooled liquids approach glass states, where molecular motion nearly halts; their volume becomes nearly invariant, yet still subject to theoretical expansion through quantum tunneling effects over extremely long timescales.
Quantum and Molecular Perspectives: The Microscopic View
At the molecular level, liquids are dynamic ensembles of atoms in constant motion. While macroscopic volume appears static, individual molecules exhibit Brownian motion, shifting positions and momentarily stretching or compressing local space. Despite these micro-scale variations, bulk volume remains stable—more a statistical emergent property than a rigid atomic constraint.Quantum mechanics deepens this complexity. Zero-point energy—the minimum energy molecules retain even at absolute zero—prevents perfect compression, preserving a baseline volume. In extreme conditions, quantum effects like tunneling allow molecules to shift positions without overcoming classical barriers, contributing to subtle, irreversible volumetric changes over time.
Quantum fluid theories further reveal that superfluids—such as liquid helium at near-zero temperatures—exhibit zero viscosity but retain a defined volume shaped by quantum coherence rather than classical forces.
Phase Transitions and Volume Sudden Shifts
Liquids undergo dramatic volumetric transformations during phase changes. Freezing water into ice reduces density by about 9%, expanding volume as hydrogen bonds lock molecules into a more open lattice. This phase shift defies the notion of constant volume.Conversely, supercritical fluids—formed when liquid and gas phases merge under high pressure and temperature—exhibit liquid-like density but gas-like diffusivity, with volume behavior intermediate between phases, yet still governed by thermodynamic principles. These transitions underscore: volume is not an immutable trait but a dynamic response to thermodynamic variables. While practical applications depend on stability, fundamental science demands a refined view of liquids—not as possessing fixed volume intrinsically, but as materials whose volume emerges from complex, condition-dependent interactions.
In essence, the answer to whether a liquid has a definite volume lies in context: under everyday conditions, volume behaves stably and predictably, reinforcing a functional definition. However, a rigorous scientific lens reveals that volume fluctuates at molecular and extreme extremes, shaped by temperature, pressure, quantum effects, and phase dynamics. Far from a rigid fact, liquid volume is a fluid truth—ever responsive, always defined less by absolutes and more by the intricate balance of forces governing matter in motion.



Related Post

Vivir Mi Vida: Marc Anthony’s Manifesto of Resilience and Identity

Delta Flight Attendants Soar High: Why Their Wages Stand Unmatched in Aviation’s Most Competitive Industry

Jesse Watters Measures Up: The Unlikely Truth Behind His Height and Public Perception

From Soul to Silver: The Echoes of Robert Ellis Silberstein and Diana Ross in the Universal Language of Music

