Breaking Down Hydrolyse ATP: The Catalyst Behind Energy Transformation in Cells
Breaking Down Hydrolyse ATP: The Catalyst Behind Energy Transformation in Cells
ATP hydrolysis stands at the heart of cellular energy dynamics—a molecular process so fundamental yet elegant that it powers nearly every life-sustaining function in living organisms. By breaking down adenosine triphosphate, cells unlock stored energy with precision and efficiency, enabling movement, synthesis, communication, and regulation. Understanding how ATP hydrolysis drives energy transformation reveals not just a chemical reaction, but a masterful biological mechanism honed by evolution.
The Molecular Architecture of ATP Hydrolysing
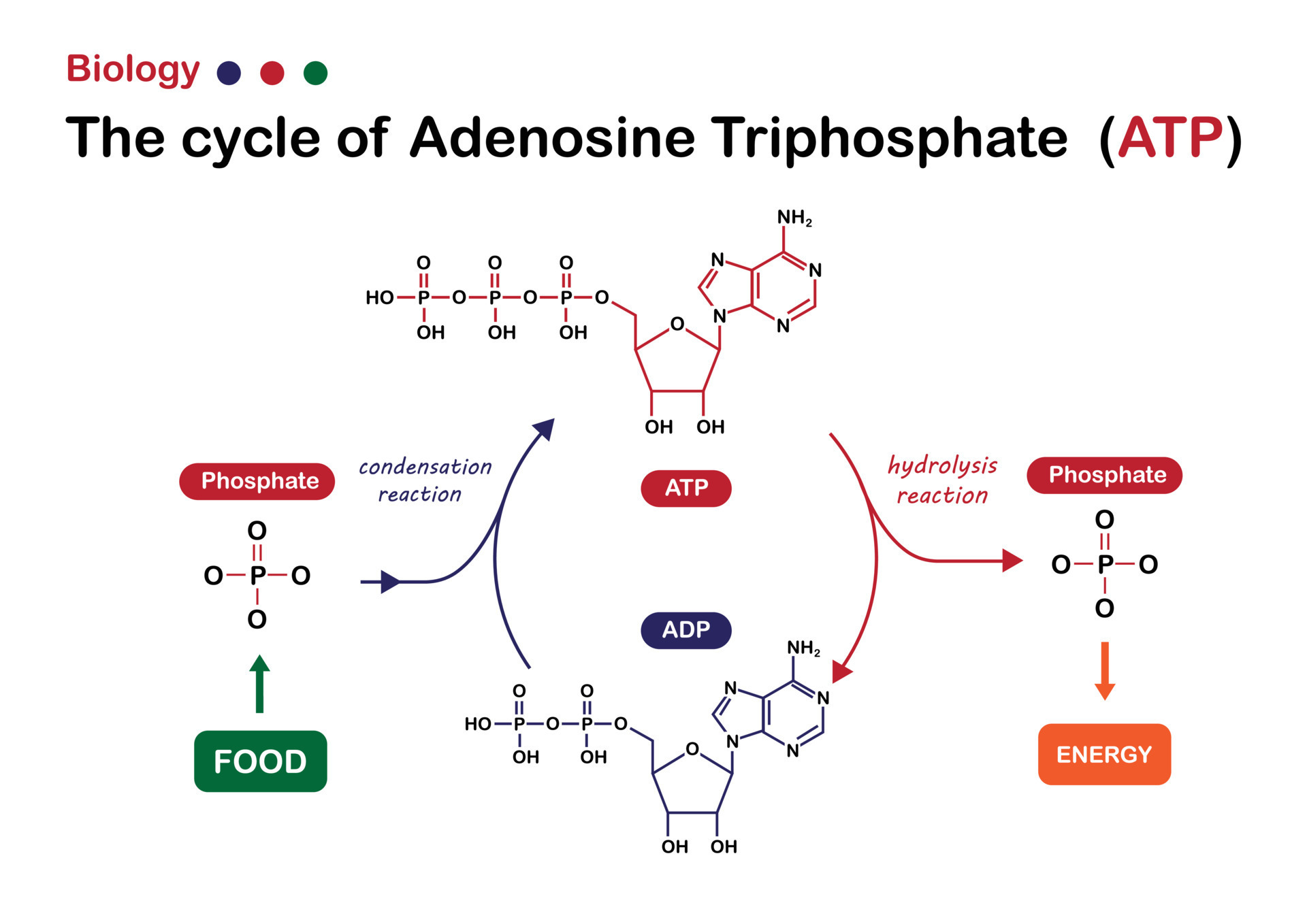
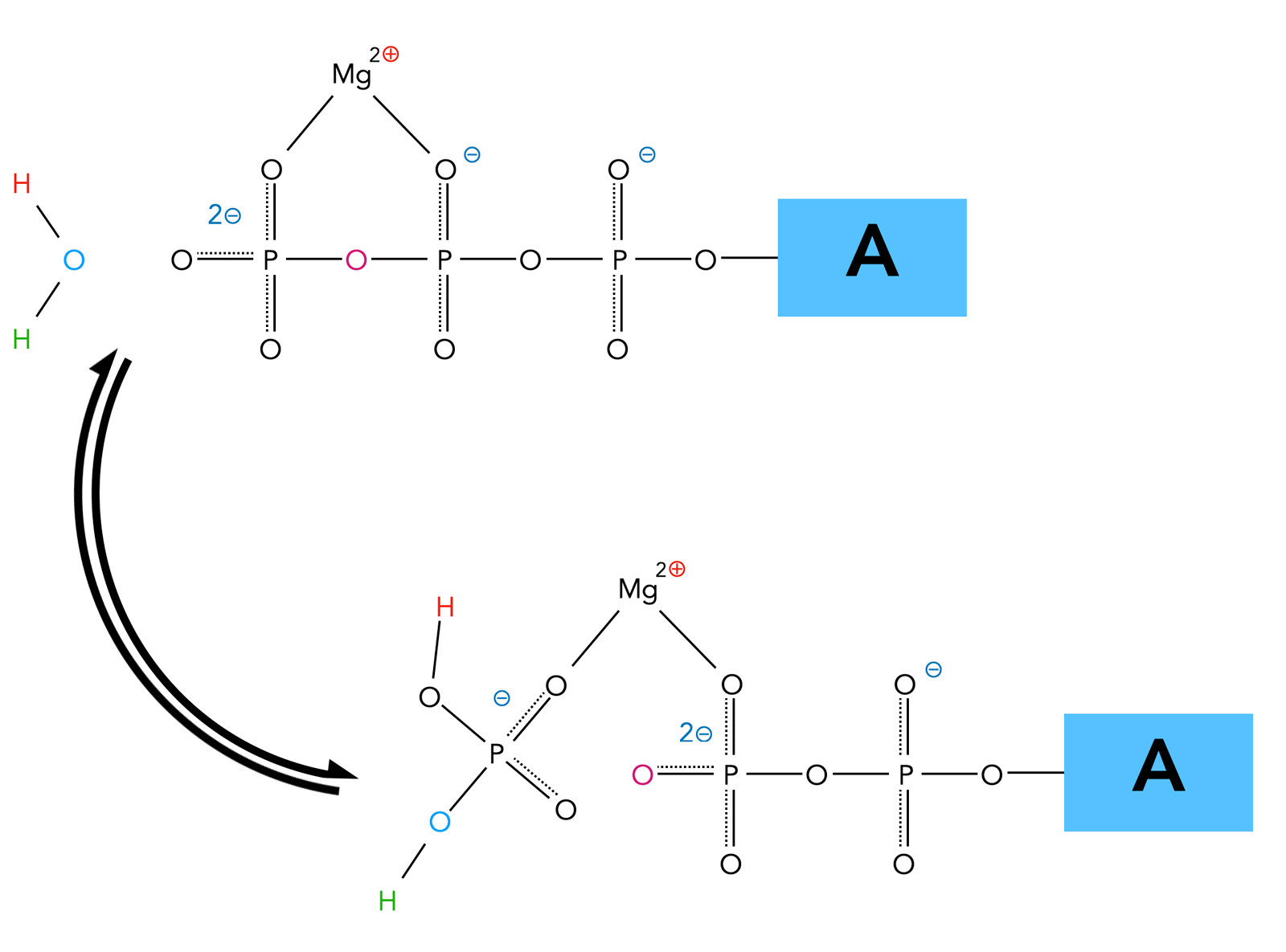
Adenosine triphosphate (ATP) serves as the primary energy currency of the cell, composed of an adenine base, a ribose sugar, and three phosphate groups linked by high-energy bonds. The terminal phosphate bond—specifically between the second and third phosphate—is the key energy reservoir. When hydrolyzed, this bond breaks to form adenosine diphosphate (ADP) and inorganic phosphate (Pi), releasing approximately 30.5 kilojoules per mole under standard conditions—a substantial yet controlled energy payload.This transformation is not random: enzyme catalysts, particularly ATPases, accelerate hydrolysis with remarkable specificity. Their active sites stabilize transition states, bind ATP tightly, and orient water molecules to facilitate bond cleavage. As Nobel laureate and biophysicist Venkatraman Ramakrishnan noted, “Enzymes transform energetics from sluggish to scalable, turning chemical potential into immediate cellular work.” Hydrolysis thus becomes a swift, regulated event—critical for timing cellular responses to environmental cues.
A Gateway to Biological Work
Hydrolysis of ATP functions as the essential bridge between energy storage and usage. The released inorganic phosphate and ADP carry this energy forward, transmitting it to other molecules via coupling reactions. For example, in muscle fibers, the hydrolysis of ATP fuels myosin motor activity, enabling contraction.In active transport, ATP hydrolysis powers pumps like the sodium-potassium (Na⁺/K⁺)-ATPase, maintaining electrochemical gradients vital for nerve signaling and cellular homeostasis. This process is elegantly efficient: instead of storing vast chemical energy in bulk, cells harness precisely timed hydrolysis to generate energy as needed. Each hydrolysis event releases energy in a manageable form—high enough to drive endergonic reactions, yet controlled to prevent wasteful overproduction.
The real beauty lies in this transformation: from stable ATP to dynamic ADP and Pi, energy becomes actionable, predictable, and non-toxic.
The Role of ATPases: Nature’s Catalytic Pioneers
ATPase enzymes are the specialized catalysts that orchestrate hydrolysis. Among them, myosin ATPase drives muscle contraction; Ca²⁺-ATPases sequester calcium to regulate muscle relaxation and cellular signaling; and ATP synthase—arguably the most sophisticated molecular machine—constructs ATP in mitochondria using proton gradients, closing the loop of energy conversion.These enzymes do far more than accelerate reactions: they ensure hydrolysis occurs at precisely the right time and place. Without ATPases, the energy stored in ATP would remain inert, trapped in molecular stasis. As biochemist Jennifer Doudna explained, “Catalysts turn potential into power—ATPases are nature’s master switchmakers of bioenergetics.” This regulation is paramount in tissues requiring rapid, coordinated energy bursts, such as neurons during synaptic transmission or cardiac muscle during contraction.
Energy Flow Networks: From Hydrolysis to Functional Output
The energy derived from ATP hydrolysis cascades through cellular networks in a finely tuned web. When ADP and Pi are regenerated—often via oxidative phosphorylation in mitochondria or glycolysis in the cytoplasm—ATP is reformed, sustaining the cycle. This dynamic equilibrium ensures energy availability matches demand, adapting swiftly to metabolic shifts.Consider photosynthesis and respiration: in chloroplasts and mitochondria alike, ATP hydrolysis and synthesis fuel electron transport chains and substrate-level phosphorylation. The cycle is continuous—not just cyclical in form, but functional in sustaining life. Each molecule of ATP, though small, is a node in a vast, intelligent network converting chemical energy into physical work, from the tiniest ion channel opening to the rhythmic beating of the heart.
Regulation: Ensuring Precision and Efficiency
Cells impose strict control over ATP hydrolysis to prevent wasteful energy leakage. Regulatory mechanisms include allosteric modulation, phosphorylation cascades, and feedback inhibition. For instance, high ATP concentrations inhibit glycolytic enzymes, slowing energy consumption when levels are ample.Conversely, low ATP—marked by elevated ADP—activates pathways to rebuild stores. Enzyme binding sites often interact with cellular signals, allowing rapid response to stress, exercise, or fasting. This precision prevents uncontrolled energy drain, preserving ATP for critical phases like neural activity or immune response.
“Precision in energy regulation separates trivial expenditure from survival-shaping reactions,” observes biochemist coincidence researcher Marie Farina. Hydrolysis is not merely a chemical event—it is a regulated, energy-smart behavior sculpted by millions of years of evolutionary refinement.
Applications Beyond the Cell: From Medicine to Biotechnology
Understanding ATP hydrolysis drives innovations across medical and technological domains.In pharmacology, targeting ATPases offers promising strategies—cancer therapies sometimes disrupt ATP-dependent pathways to halt tumor growth. Neurodegenerative disease research investigates ATP depletion as a hallmark of neuronal fatigue and degeneration. Meanwhile, synthetic biology mimics natural ATP cycling to engineer biofuels and bioelectronics, harnessing energy transformation at the molecular scale.
In diagnostics, measuring ATP hydrolysis rates provides insights into metabolic health, oxidative stress, and cellular viability. Fluorescent ATP sensors, for instance, map energy dynamics in live cells, revealing real-time responses to drugs or environmental toxins. These tools deepen understanding and accelerate development of targeted interventions.
Hydrolysis of ATP is far more than a biochemical footnote—it is the foundational engine of cellular life. This precise, enzyme-facilitated transformation converts stored chemical potential into the dynamic energy required for growth, movement, communication, and resilience. As scientific inquiry advances, unraveling the subtleties of ATP hydrolysis continues to deepen our grasp of biology’s most essential processes, revealing nature’s brilliance in energy’s elegant manipulation.


Related Post

Debra Winger’s Battle: Brain Hemorrhage and the Resilience of a Hollywood Icon

Breaking News: Bandung Reacts in Reverence as Solemn Condolences Flow Through the City
Haley Van Lith’s Boyfriend Sparked a Cultural Conversation: Influence, Identity, and Heartfelt Connection

Mackenzie Davis Relationships A Deep Dive Into Love, Personal Growth, and Compatibility

